Sewage Pathogens & Contaminants in Fruits and Vegetable
Sewage Pathogens & Contaminants in Fruits and Vegetable
- POST a QUESTION or COMMENT about fruits & vegetables produced from soils irrigated with septic wastewater or septage
Research on Sewage Pathogens & Contaminants in Fruits and Vegetables:
Results of a study of health hazards of consuming fruits and vegetables that may contain contaminants produced from irrigation with septic effluent.
InspectAPedia tolerates no conflicts of interest. We have no relationship with advertisers, products, or services discussed at this website.
Bacterial pathogens recovered from vegetables irrigated by wastewater in Morocco
"Bacterial Pathogens Recovered from Vegetables Irrigated by Wa stewater in Morocco", Y. Karamoko, K. Ibenyassine, M. M. Ennaji, B. Anajjar, R. Ait Mhand, M. Chouibani, Journal of Environmental Health, June 2007. This article has also been re-published by thefreelibrary.com.
At our home page on this topic, SEWAGE CONTAMINANTS in FRUIT / VEGETABLES, we pose a series of questions regarding the article below, and we suggest some follow-up research. We also express an opinion about the comparative pathogenic contamination risks identified by this research article and the risks likely to be faced by growing fruits or vegetables over or near septic drainfields.
Abstract
The authors obtained 50 vegetable samples from various regions in Morocco and examined them to determine the micro biological quality of these products. Aerobic count, coliform, enterococci, and Staphylococcus areus were evaluated. This analysis revealed high levels of enterococci, fecal coliforms, and total coliforms.
No coagulase-positive Staphylococcus aureas was detected in any of the samples analyzed.
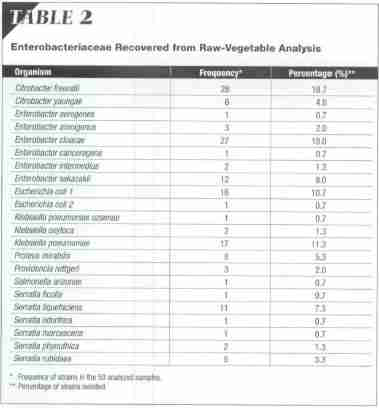
Biochemical identification of Enterobacteriaceae showed the presence of Citrobacter freundii (28 percent), Enterobacter cloacae (27 percent), Escherichia coli (16 percent), Enterobacter sakazakii (12 percent), Klebsiella pneamoniae (17 percent), Serratia liquefaciens (11 percent), and Salmonella arizonae (0.7 percent).
The results clearly demonstrate that vegetables irrigated with untreated wastewater have a high level of microbiological contamination. Consequently, these vegetables may be a threat for the Moroccan consumer and may be considered a serious risk to Moroccan public health.
No warranty is given about the accuracy of the copy. Users should refer to the original published version of the material for the full abstract. (See the original article citation in the Journal of Environmental Health, June 2007 listed above and at the end of this page.)
SEPTIC SYSTEMS - INTRODUCTION
An increase in consumption of fresh fruits and vegetables worldwide has been paralleled by an increase in the number of foodborne illnesses attributed to fresh products. Numerous reports have indicated that raw vegetables may harbor potential foodborne pathogens (Beuchat, 1996).
In particular, tomatoes, cantaloupes, and sprouts have been linked to outbreaks of salmonellosis ( Guo, Chen, Brackett, & Beuchat, 2001 ), and outbreaks of illnesses caused by Escherichia coli O157:H7 have been associated with melon, apple cider, lettuce, and radish sprouts ( Breuer et al., 2001 ).
Moreover, coleslaw, cabbage, potatoes, radishes, bean sprouts, and cucumbers contaminated with Listeria monocytogenes have been linked to disease outbreaks ( Shearer, Strapp, & Joerger, 2001 ), and salad vegetables also may be contaminated with Campylobacter Evans, Ribeiro, & Salmon, 2003 ).
In Morocco, vegetable products have been in great demand in recent years. Since the rate of precipitation has been very low during these last decades, wastewater is increasingly being used in agriculture.
Little information is available on the number of human foodborne-illness outbreaks that have occurred from consumption of raw vegetables. The use of raw sewage to irrigate crops is an important mechanism that helps to propagate conditions conducive to cholera and typhoid fever ( Castro-Rosas & Escartin, 2000 ). Increases in foodborne illnesses during the summer are not fully understood, although fresh produce likely plays a role since it is consumed in higher quantities during the summer.
The study reported here investigated the occurrence of pathogenic bacteria in vegetables irrigated by untreated wastewater in Morocco. Irrigated vegetables do not undergo any control before being exposed in the markets, after which they may be eaten cooked or raw. The purpose of the study was 1) to determine the bacterial quality of vegetables irrigated with untreated wastewater, 2) to sensitize farmers to the dangers from use of untreated wastewater for irrigation, and 3) to elucidate the risk to Moroccan public health.
Materials and Methods
Samples
A total of 50 vegetable samples were procured for bacteriological examination. Vegetables of various types were obtained from several wastewater-irrigated agricultural regions in Morocco. Sampling was conducted from August 2002 to July 2004. The vegetable samples were collected in sterile polyethylene bags, and steps were taken to avoid contamination of the vegetables by soil or other contamination sources. Each sample was collected in triplicate to prevent sampling error. The vegetables were tomato, radish, cucumber, eggplant, potato, pepper, garden pea, gourd, zucchini, artichoke, broad bean, turnip, onion, French bean, and lettuce.
Vegetables of various types were obtained from several wastewater-irrigated agricultural regions in Morocco.
Sampling was conducted from August 2002 to July 2004. The vegetable samples were collected in sterile polyethylene bags, and steps were taken to avoid contamination of the vegetables by soil or other contamination sources. Each sample was collected in triplicate to prevent sampling error. The vegetables were tomato, radish, cucumber, eggplant, potato, pepper,
All the samples were transported to the laboratory under low temperature (<7[degrees]C) and stored at 4[degrees]C until testing. They were analyzed within 20 hours of sampling. Each sample was rinsed several times with sterile distilled water to eliminate the soil. Before analysis, 25 g of each sample was homogenized for two minutes with 225 mL of 0.1 percent sterilepeptone water with a Model 400 Stomacher (Seward Medical, London) and serially diluted.
Bacteriological Analysis
Using the spread-plate technique and 100 [micro]L from the serial dilution, the authors prepared duplicate plates for the determination of aerobic plate counts (APC) Enterobactericiceae, fecal coliforms, total colilorms, Staphylococcus,and Streptococcus.
Aerobic plate counts were made with plate count agar. (Merck), and plates were incubated at 30[degrees]C for 48 hours. Then all colonies on plates were counted. Enterococci counts were made with Slanetz and Bartley agar (Biokar).
The plates were incubated at 37[degrees]C for 48 hours, and all typical colonies (pink or dark red with a narrow whitish border) were counted. For the coliform counts, violet red bile agar (from Merck) was used for direct plating, and plates were incubated at 37[degrees]C for 24 hours and 42C for total coliforms and fecal coliforms, respectively.
Typical colonies were round, red to pink, 0.5 to 2 mm in diameter, and surrounded with a red-to-pink halo. Staphlococcus aureus counts were determined with Baird-Parker Agar (Difco) with egg yolk--tellurite emulsion, and plates were incubated at 37[degrees]C for 24 hours to 48 hours.
Colonies selected from the agar surface were examined under microscope for Gram stain and were tested for catalase reaction and then for coagulase activity with plasma rabbit (Biokar).
To isolate Salmonella spp., we pummeled a 25-g sample in a stomacher with 225 mL of buffered peptone water and pre-enriched the homogenate 37[degrees]C for 18 hours. A 100-[micro]L sample was subcultured into 10 mL of Rappaport Vassiliadis Broth (Difco) and enriched at 41.5[degrees]C for 24 hours and 48 hours.
One mL of the pre-enrichment broth was simultaneously inoculated into 10 mL of selenite cysteine broth and enriched at 37[degrees]C for 22 hours and 48 hours. Both enrichment broths were streaked onto xylose lysine deoxycholate agar (Merck) and Salmonella-Shigella agar, and incubated at 37[degrees]C for 22 hours.
For selective plating, presumptive Salmonella colonies from selective plates were confirmed with the API 20E identification system (BioMerieux). The Enterobacteriaceae strain was isolated with Levine-EMB agar (Merck). The plates were incubated at 37[degrees]C for 18 hours, and colonies growing on the plates were examined under a microscope for Gram stains and tested for catalase and oxydase reactions. For identification of all strains, the API 20E identification system (BioMerieux) was used.
Results
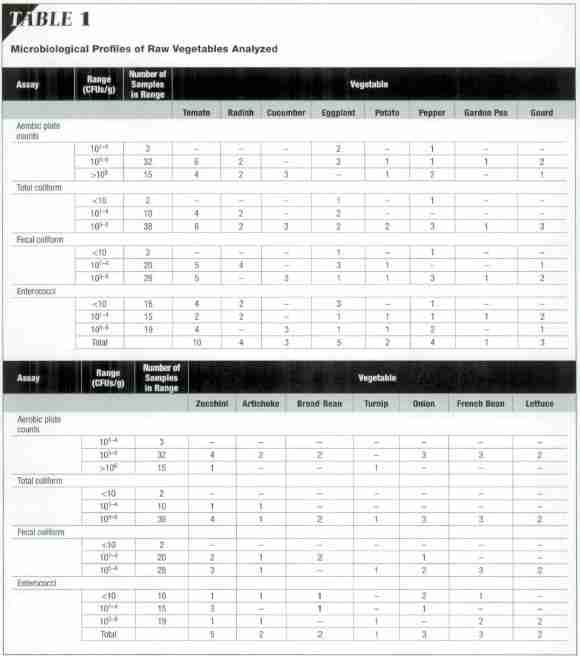
These analyses showed high aerobic-plate, total-coliform, fecal-coliform, and enterococci counts. Coagulase-positive Staphylococcus aureus was not detected in any samples (Table 1).
The frequencies with which the bacteria were recovered from samples are given in Table 2. Citrobacter freundii and Enterobacter cloacae were recovered most frequently (from 28 percent of samples). Other Gramnegative bacteria that were frequently isolated were Escherichia coli (16 percent), Enterobacter sakazakii (12 percent), Klebsiella pneumoniae (17 percent), and Serratia liquejaciens (11 percent).
Discussion
Foodborne diseases remain an important public health threat worldwide, and one of the most important food safety hazards is associated with raw vegetables. The large number of total microorganisms and fecal-contamination indicators (E. coli, coliform, and enterococci) detected in the vegetable samples we surveyed indicates a potential health hazard to consumers. Madden has discussed potential sources of microbial contamination of fresh fruit and vegetables during growth, harvest, distribution, and processing (1992).
The bacteria that the authors found on samples belonged most frequently to the Citrobacter-Enterobacter-Serratia group of Enterobacteriaceae. Although usually regarded as human pathogens, these members of Enterobacteriaceae family have also been recognized as inhabitants of soil and plants (Wright, Kominos, & Yee, 1976).
Thus, vegetables may serve as a reservoir from which the bacteria named above can colonize and infect a susceptible host.
In many countries, urban wastewater is used to irrigate agricultural land. This way of disposing of urban sewage water has several advantages. Wastewater contains a lot of nutrients, which increase crop yields without use of fertilizer. Furthermore, sewage water is an alternative water source in arid and semi-arid areas where water is scarce. Some disadvantages are that wastewater can contain heavy metals, organic compounds, and a wide spectrum of enteric pathogens that may have a negative impact on the environment and human health.
The study reported here demonstrates that a potential for disease transmission exists when wastewater is used for irrigation. Pathogens that have been transported by wastewater can survive in soil or on crops.
The actual risk of disease transmission, however, is related to whether this survival time is long enough to allow transmission to a susceptible host. The crop and the field are the link between the pathogen in the wastewater and the potential for infection.
The factors controlling transmission of disease are agronomic examples of such factors are the crop grown, the irrigation method used to apply wastewater, and cultural and harvesting practices.
Consumption of salad irrigated by wastewater has been found to be responsible for shigellosis in England (Frost, McEvoy, Bentley, Andersson, & Rowe, 1995). Numerous opportunities exist for attachment and penetration of pathogenic bacteria into lettuce in the field, as well as during harvesting, processing, and marketing, especially when a contaminated product is exposed to water or is damaged (Takeuchi et al., 2001).
Guo and co-authors have demonstrated that soil and water are potential reservoirs from which Salmonella can contaminate tomatoes (Guo, Chen, Brackett, & Beuchat, 2002). The pathogen can survive in most soils in high numbers for at least 45 days and can infiltrate the tissues of tomatoes during contact with inoculated soil.
This mechanism may explain the discovery of Salmonella arizonae on tomatoes in the investigation reported here. The hydroponics system used in another study provided a controlled environment in which to study the possible association of Salmonellae with aerial tissues, with minimal concerns about environmental contamination or temperature fluctuation (Guo, Iersel, Chen, Brackett, & Beuchat, 2002).
That study provided evidence that Salmonellae can be transported from an inoculated nutrient solution to the hypocotyls, cotyledons, stems, and leaves of young tomato plants. In addition, the work of Guo and co-authors (2001) revealed the ability of Salmonella to survive on or in tomato fruits throughout the course of plant growth, flowering, fruit development, and fruit maturation (2001).
Another study showed that Escherichia coli O157:H7 may be present not only on outer surfaces, but also in the inner tissues and stomata of cotyledons of radish sprouts grown from seeds experimentally contaminated with the bacterium (Itoh et al., 1998). Indeed, this mechanism can be inferred from the fact that enteric pathogens were isolated from inside vegetables during our investigation.
Numerous foodborne diseases caused by Salmonella enterica and Escherichia coli O157:H7 have been associated with contaminated alfalfa clover, and bean sprouts (Puohiniemi, Heiskanen, & Siitonen, 1997). Constant moisture, nutrients released by the sprouting seeds, and warm temperatures are conducive to the growth of human bacterial pathogens such as S. enterica and E. coli O157:H7 (Charkowski, Barak, Sarreal, & Mandrell, 2002).
The fact that some bacteria and not others are present can be explained by a difference in their capacity to attach to plants. For example, Salmonella enterica attaches as well as plant-associated bacteria and significantly better than E. coli to alfalfa sprouts (Barak, Whitehand, & Charkowski, 2002).
Conclusion
The authors' study demonstrated that the vegetables analyzed may be an important source of foodborne bacterial-illness outbreaks in humans, especially when the vegetables are consumed raw. In addition, vegetables of these types can also be a vehicle for Giardia cysts and Ascaris eggs (Amahmid, Asmama, & Bouhoum, 1999). To protect public health, the use of raw sewage in the irrigation of the vegetable culture must be prohibited.
So it is necessary to consider the generalized program of wastewater treatment in Morocco. Moreover, the requirements for treated wastewater must respect the sanitary standards for agricultural reuse. There is an urgent need for development and validation of standard methods of eliminating the pathogenic microorganisms from raw vegetables.
Acknowledgements: This work was financially supported by the PARS and PROTARS programs of the Moroccan National Research Council. The authors acknowledge Hassan Tajnari (Service for Protection of Plants, Ministry for Agriculture, Marrakech-Morocco) and Meryem Aakill (Division of Plant Health Control, Ministry for Agriculture, Rabat-Morocco) for their assistance during sampling and for their technical help.
Corresponding Author: M.M. Ennaji, Professor, Laboratory of Virology and Hygiene & Microbiology, Department of Biology, Faculty of Science and Technology, University Hassan II-Mohammedia, Mohammedia, Morocco 20650. E-mail: m.ennaji@univh2m.ac.ma.
Useful Research Citations on Sewage Contamination of Food Crops Grown In or Over Septic Fields
- Al-Hamaiedeh, H., and M. Bino. "Effect of treated grey water reuse in irrigation on soil and plants." Desalination 256, no. 1 (2010): 115-119.
Abstract:
The use of treated grey water (GW) for irrigation in home gardens is becoming increasingly common in Jordan. In this study treated GW produced from 4-barrel and confined trench (CT) treatment units were used for irrigation of olive trees and some vegetable crops. The quality of treated and untreated GW was studied to evaluate the performance of treatment units and the suitability of treated GW for irrigation according to Jordanian standard.
Effect of treated GW reuse on the properties of soil and irrigated plants at Al-Amer villages, Jordan, has been investigated. The results showed that salinity, sodium adsorption ratio (SAR), and organic content of soil increased as a function of time, therefore leaching of soil with fresh water was highly recommended. The chemical properties of the irrigated olive trees and vegetable crops were not affected, while the biological quality of some vegetable crops was adversely affected. - Dar, Imran A., Mithas A. Dar, and K. Sankar. "Nitrate contamination in groundwater of Sopore town and its environs, Kashmir, India." Arabian Journal of Geosciences 3, no. 3 (2010): 267-272.
Excerpt from abstract: "... The hydrochemical data of 15 samples indicates that the concentration of almost all parameters fall within the permissible limits except nitrate."
An attempt has been made in this research work to evaluate the concentration of nitrate in groundwater and its management in Apple town and its environs. Groundwater pollution has been reported in many aquifers because of high concentration of nitrate in ground water, which is the result of excessive use of fertilizers to cropland.
Systematic sampling was done, with a view to understand the source of nitrate concentration in the study area. Fifteen sample sites were selected and the samples were taken for a baseline study to understand the geochemistry of the study area and to assess its physicochemical characteristics.
The water quality parameters were investigated for summer (May, 2007) and winter (December, 2007) seasons and were compared with the standard values given by ICMR / WHO. The hydrochemical data of 15 samples indicates that the concentration of almost all parameters fall within the permissible limits except nitrate. Linear Trend Analysis on seasonal and annual basis clearly depicted that nitrate pollution in the study area is increasing significantly. About 85% of samples during summer season and 67% of the samples during winter season were showing a high concentration of nitrate, exceeding permissible limit of WHO (50 mg/l), which is due to the use of nitrogenous fertilizers in the study area. Appropriate methods for improving the water quality and its management in the affected areas have been suggested.
تلوث المياه الجوفية بالنترات في مدينة سوبور ومحيطها بمقاطعة بارامولا كشمير – الهنديهدف هذا البحث إلى تقييم تركيز النترات في المياه الأرضية وإدارتها في مدينة سوبور ومحيطها. قد لوحظ تلوث المياه الجوفية في كثير من الخزانات الجوفية نتيجة للتركيز العالي للنترات في المياه الأرضية وذلك للاستخدام الجائر للأسمدة في زراعة المحاصيل الزراعية.في هذا الخصوص تم تصميم منهجية أخذ العينات بغرض تحديد مصدر النترات بمنطقة الدراسة. و تم أخذ 15 عينة لدراسة وللتعرف على الكيمياء الأرضية بمنطقة الدراسة وتحليل الخواص الكيميائية الطبيعية. تم فحص عناصر جودة المياه لصيف 2007 (شهر مايو) وشتاء 2007 (شهر ديسمبر) وتم مقارنتهم بالمعايير الخاصة ب ICMR/WHO ودلت البيانات الهيدروكيميائية للـ 15 عينة أن جميع المعاملات تقع في الحدود المسموح بها ماعدا النترات. ودلت نتائج التحليل الخطى على المستوى الفصلي والسنوي أن تلوث النترات يزداد بدرجة ملحوظة. كما دلت 85% من عينات الصيف و 65% من عينات الشتاء على تركيزات عالية للنترات تفوق الحدود المسموح بها من ICMR/WHO (50 مليجرام/لتر)، وقد يُعزى ذلك إلى استخدام المسمدات النيتروجينية. وقد تم اقتراح طرق مناسبة لتحسين نوعية المياه وإدارتها بمنطقة الدراسة. - Heinonen-Tanski, Helvi, and Christine van Wijk-Sijbesma. "Human excreta for plant production." Bioresource technology 96, no. 4 (2005): 403-411.
This study explores use of human excreta as a natural resource for agriculture. - Heukelekian, H. "Utilization of sewage for crop irrigation in Israel." Sewage and Industrial Wastes (1957): 868-874.
The authors argue that
Failures and difficulties with the disposal of sewage by crop irrigation need not arise when utilization is the main objective. The natural tendency with land disposal of sewage is to apply as much sewage as possible in a limited area at rates higher than can percolate through the soil. Waterlogging of the soil and odors are the common result of such practice. The anaerobic condition which may result is known as "soil sickness".
On the other hand, when the application of sewage to the land is geared to the needs of the soil and the crop, odor and waterlogging difficulties do not arise. There are a number of examples all over the world of successful utilization of sewage for crop irrigation.
The aricle continues to point out important variables including the level of rainfall, even when sewage application rate is controlled to be suitable for crops. - Illinois Windbreak Manual, A. Field. "Illinois." (1990). [PDF] - http://web.extension.illinois.edu/forestry/iwm_complete.pdf
- Leff, Jonathan W., and Noah Fierer. "Bacterial communities associated with the surfaces of fresh fruits and vegetables." PloS one 8, no. 3 (2013): e59310.
- Abstract:
Fresh fruits and vegetables can harbor large and diverse populations of bacteria. However, most of the work on produce-associated bacteria has focused on a relatively small number of pathogenic bacteria and, as a result, we know far less about the overall diversity and composition of those bacterial communities found on produce and how the structure of these communities varies across produce types.
Moreover, we lack a comprehensive view of the potential effects of differing farming practices on the bacterial communities to which consumers are exposed. We addressed these knowledge gaps by assessing bacterial community structure on conventional and organic analogs of eleven store-bought produce types using a culture-independent approach, 16 S rRNA gene pyrosequencing.
Our results demonstrated that the fruits and vegetables harbored diverse bacterial communities, and the communities on each produce type were significantly distinct from one another. However, certain produce types (i.e., sprouts, spinach, lettuce, tomatoes, peppers, and strawberries) tended to share more similar communities as they all had high relative abundances of taxa belonging to the family Enterobacteriaceae when compared to the other produce types (i.e., apples, peaches, grapes, and mushrooms) which were dominated by taxa belonging to the Actinobacteria, Bacteroidetes, Firmicutes, and Proteobacteria phyla.
Although potentially driven by factors other than farming practice, we also observed significant differences in community composition between conventional and organic analogs within produce types. These differences were often attributable to distinctions in the relative abundances of Enterobacteriaceae taxa, which were generally less abundant in organically-grown produce.
Taken together, our results suggest that humans are exposed to substantially different bacteria depending on the types of fresh produce they consume with differences between conventionally and organically farmed varieties contributing to this variation. - Mara, Duncan. "Sanitation: What's the Real Problem?." IDS Bulletin 43, no. 2 (2012): 86-92.
Abstract:
The vast number of people without sanitation raises the question why this is so. It cannot be a lack of adequate sanitation technologies as these exist for all situations from dispersed rural communities to high-density low-income urban areas. Nor cannot it be money as development banks will readily fund a well-prepared sanitation proposal. The real sanitation problem must surely lie with those developing-country governments who have shown little commitment in practice to sanitation despite international sanitation advocacy since 1980. Their lack of commitment is clearly shown in the number of ‘open defecators’ in the world today. There are fortunately some countries that have done well: Malaysia, Thailand and Vietnam, for example, but they are a clear minority. - Mogheir, Y., T. Abu Hujair, Z. Zomlot, A. Ahmed, and D. Fatta. "Treated Wastewater Reuse in Palestine." In International conference Water Value and Rights, Ramallah-Al-Bireh, Palestine May. 2005.
Abstract:
Treated wastewater resource is an environmental, social and economical good that needs to be managed in appropriate way. Palestinian Territories, as in most of the neighboring countries in the Middle East region, acknowledges the importance of this resource in improving the water deficit by reusing the treated wastewater in agricultural production, industrial sector and recharges the aquifer.
However, this resource is strictly sensitive and has adverse impacts on the public health. Both negative and positive impacts of the treated wastewater resource should be considered. Therefore, Environment Quality Authority (EQA) has prepared Environmental Law and Standards Draft for reusing the treated wastewater.
Moreover, Palestinian Water Authority (PWA) prepared a National Water Plan where the treated wastewater reuse has an essential part. EQA is currently sharing a project which considers the development of tools and guidelines for the promotion of the sustainable urban wastewater treatment and reuse in the agricultural production in the Mediterranean countries.
This paper presents the Palestinian practices and plans in wastewater treatment and reuse. - Nair, Jaya. "Wastewater garden--a system to treat wastewater with environmental benefits to community." Water Science & Technology 58, no. 2 (2008).
Abstract:
Many communities and villages around the world face serious problems with lack of sanitation especially in disposing of the wastewater--black water and grey water from the houses, or wash outs from animal rearing sheds. Across the world diverting wastewater to the surroundings or to the public spaces are not uncommon.
This is responsible for contaminating drinking water sources causing health risks and environmental degradation as they become the breeding grounds of mosquitoes and pathogens.
Lack of collection and treatment facilities or broken down sewage systems noticed throughout the developing world are associated with this situation. Diverting the wastewater to trees and vegetable gardens was historically a common practice.
However the modern world has an array of problems associated with such disposal such as generation of large quantity of wastewater, unavailability of space for onsite disposal or treatment and increase in population.
This paper considers the wastewater garden as a means for wastewater treatment and to improve the vegetation and biodiversity of rural areas. This can also be implemented in urban areas in association with parks and open spaces.
This also highlights environmental safety in relation to the nutrient, pathogen and heavy metal content of the wastewater. The possibilities of different types of integration and technology that can be adopted for wastewater gardens are also discussed. - Nelson, M., F. Cattin, M. Rajendran, and L. Hafouda. "Value-Adding through Creation of High Diversity Gardens and Ecospaces in Subsurface Flow Constructed Wetlands: Case Studies in Algeria and Australia of Wastewater Gardens Systems." In Proceedings of 11th International Conference on Wetland Systems for Water Pollution Control, edited by S. Billore, P. Dass, and J. Vymazal, vol. 1, pp. 344-356. Ujjain, India: Institute of Environmental Management and Plant Sciences, Vikram University, 2008.
- Sipahioglu, H. M., A. Myrta, N. Abou‐Ghanem, B. Terlizzi, and V. Savino. "Sanitary status of stone‐fruit trees in East Anatolia (Turkey) with particular reference to apricot." EPPO Bulletin 29, no. 4 (1999): 439-442.
Abstract:
Field surveys were carried out in the main stone-fruit-growing areas of East Anatolia (Turkey) to assess the sanitary status of varietal collections, mother blocks and commercial orchards. The presence of virus and virus-like diseases was ascertained by enzyme-linked immunosorbent assay (ELISA), sap transmission to herbaceous hosts, graft transmission to peach cv. GF305 and molecular hybridization tests.
A total of 1019 samples was tested by ELISA (859 apricot, 120 cherry, 21 almond and 19 peach). The sanitary status of apricot was extremely satisfactory, as the infection level was less than 0.3%. Cherry and almond, however, showed 21% and 33% infection respectively.
The viruses identified were apple chlorotic leaf spot trichovirus (ACLSV), prune dwarf ilarvirus (PDV) and prunus necrotic ringspot ilarvirus (PNRSV).
The commonest virus was PDV. Plum pox potyvirus (PPV), apple mosaic ilarvirus (ApMV) and the nepoviruses tomato black ring (TBRV), raspberry ringspot (RpRSV), strawberry latent ringspot (SLRV), cherry leaf roll (CLRV), arabis mosaic (ArMV) and tomato ringspot (ToRSV) were not encountered. Peach latent mosaic viroid (PLMVd) and hop stunt viroid (HSVd) were not detected either. - Watson, John Lionel Airay. "OXIDATION PONDS AND USE OF EFFLUENT IN ISRAEL." In ICE Proceedings, vol. 22, no. 1, pp. 21-40. Thomas Telford, 1962.
Excerpt from abstract:
The evolution of sewage treatment in oxidation ponds in Israel overthe past decade is described. The high degree of purification is shown to be due to the work of algae, of which Chlorella is the most prolific. The photo-synthetic action of light and the heat that it generates build new algal cell material...
Septic & Sewage Pathogens and Contaminants, References & Research Articles
- Amahmid, O., Asmama, S., & Bouhoum, K. (1999). The effect of waste water reuse in irrigation on the contamination level of food crops by Giardia cysts and Ascaris eggs. International Journal of Food Microbiology, 49(1-2), 19-26.
- Barak, J.D., Whitehand, L.C., & Charkowski, A.O. (2002). Differences in attachment of Salmonella enterica serovars and Escherichia coli O157:H7 to alfalfa sprouts. Applied and Environmental Microbiology, 68(10), 4758-4763.
- Beuchat, L.R. (1996). Pathogenic microorganisms associated with fresh produce. Journal of Food Protection, 59(2), 204-216.
- Breuer, T., Benkel, D.H., Shapiro, R.L., Hall, W.N., Winnett, M.M., Linn, M.J., Timothy, J.N., Barrett, J., Dietrich, S., Downes, F.P., Toney, D.M., Pearson, J.L., Rolka, H., Slutsker. L., & Griffin, P.M. (2001). A multi-state outbreak of Escherichia coli O157:H7 infections linked to alfalfa sprouts grown from contaminated seeds. Emerging Infectious Diseases, 7(6), 977-982.
- Castro-Rosas, J., & Escartin, E.F. (2000). Survival and growth of Vibrio cholerae O1, Salmonella typhi, and Escherichia coli O157:H7 in alfalfa sprouts. Journal of Food Science, 65(1), 162-165.
- Charkowski, A.O., Barak, J.D., Sarreal, C.Z., & Mandrell, R.E. (2002). Growth and colonization patterns of Salmonella enterica and Escherichia coli O157:H7 on alfalfa sprouts and the effects of sprouting temperature, i inoculum /in·oc·u·lum/ ( -ok´u-lum ) pl. inoc´ula material used in inoculation.
- Evans, M.R., Ribeiro, C.D., & Salmon, R.L. (2003). Hazards of healthy living: Bottled water and salad vegetables as risk factors for Campylobacter infection. Emerging Infectious Disease, 9(10), 1219-1225.
- Frost, J.A., McEvoy, M.B., Bentley, C.A., Andersson, Y., & Rowe, B. (1995). An outbreak of Shigella sonnei infection associated with consumption of iceberg. Emerging Infectious Disease, 1(1), 26-28.
- Guo, X., Chen, J., Brackett, R.E., & Beuchat, L.R. (2001). Survival of Salmonellae on and in tomato plants from the time of inoculation at flowering and early stages of fruit development through fruit ripening, said of meat. See curing. . Applied and Environmental Microbiology, 67(10), 4760-4764.
- Guo, X., Chen, J., Brackett, R.E., & Beuchat, L.R. (2002). Survival of Salmonellae on tomatoes stored at high relative humidity, in soil, and on tomatoes in contact with soil. Journal of Food Protection, 65(2), 274-279.
- Guo, X., Iersel, M.W.V., Chen, J., Brackett, R.E., & Beuchat, L.R. (2002). Evidence of association of salmonellae with tomato plants grown hydroponically in inoculated nutrient solution. Applied and Environmental Microbiology, 68(7), 3639-3643.
- Itoh, Y., Sugita-Konishi, Y., Kasuga, E, Iwaki, M., Hara-Kudo, Y., Saito, N., Noguchi, Y, Konuma, H., & Kumagai, S. (1998) Enterohemorrhagic Escherichia coli enterohemorrhagic Escherichia EHEC Any of the E coli serotypes–eg O29, O39, O145 that produces shiga-like toxins, causing bloody inflammatory diarrhea, evoking a HUS. See Escherichia coli O157:H7, Hemolytic uremic syndrome. O157:H7 present in radish sprouts. Applied and Environmental Microbiology, 64(4), 1532-1535.
- Madden, J.M. (1992). Microbial pathogens in fresh produce--The regulatory perspective. Journal of Food Protection, 55, 821-823.
- McMahon, M.A.S., & Wilson, I.G. (2001). The occurrence of enteric pathogens and Aeromonas species in organic vegetables. International Journal of Food Microbiology, 70(1-2),155-162.
- Puohiniemi, R., Heiskanen, T., & Siitonen, A. (1997). Molecular epidemiology of two international sprout-borne Salmonella outbreaks. Journal of Clinical Microbiology . 35(10), 2487-2491.
- Shearer, A.E., Strapp, C.M., & Joerger, R.D. (2001). Evaluation of polymerase chain reaction-based system for detection of Salmonella enteritidis, Escherichia coli O157:H7, Listeria spp., and Listeria monocytogenes on fresh fruit and vegetables. Journal of Food Protection, 64(6), 788-795.
- Takeuchi, K., Hassan, A.N., & Frank, J.F. (2001). Penetration of Escherichia coli O157:H7 into lettuce as influenced by modified atmosphere and temperature. Journal of Food Protection, 64(11), 1820-1823.
- Wright, C., Kominos, S.D., & Yee, R.B. (1976). Enterobacteriaceae and Pseudomonas aeruginosa recovered from vegetable salads. Applied and Environmental Microbiology, 31(3), 453-454.
Although most of the information presented in the Journal refers to situations within the United States, environmental health and protection know no boundaries.
The Journal periodically runs International Perspectives to ensure that issues relevant to our international constituency, representing over 60 countries worldwide, are addressed. Our goal is to raise diverse issues of interest to all our readers, irrespective of origin.
K. Ibenyassine, D.E.S.A.
R. Ait Mhand, Ph.D.
Y. Karamoko, Ph.D.
B. Anajjar, Ph.D.
M. Chouibani
M.M. Ennaji, Ph.D.
...
Continue reading at SEWAGE CONTAMINANTS in FRUIT / VEGETABLES or select a topic from the closely-related articles below, or see the complete ARTICLE INDEX.
Or see
- BACTERIAL PATHOGENS in FRUIT & VEGETABLES - research citations on sewage contaminants in fruits & vegetables
- GARDENS NEAR SEPTIC SYSTEMS
- VEGETABLE or PRODUCE DISINFECTION
- SEPTIC SYSTEMS, PLANTS OVER - risk of tree or plant damage to septic drainfields
- SEWAGE CONTAMINANTS in FRUIT / VEGETABLES
- SEWAGE NITROGEN CONTAMINANTS
- SEWAGE PATHOGENS in SEPTIC SLUDGE for a list and discussion of of the common pathogens and other contaminants in residential sewage
Suggested citation for this web page
BACTERIAL PATHOGENS in FRUIT & VEGETABLES at InspectApedia.com - online encyclopedia of building & environmental inspection, testing, diagnosis, repair, & problem prevention advice.
Or see this
INDEX to RELATED ARTICLES: ARTICLE INDEX to SEPTIC SYSTEMS
Or use the SEARCH BOX found below to Ask a Question or Search InspectApedia
Ask a Question or Search InspectApedia
Try the search box just below, or if you prefer, post a question or comment in the Comments box below and we will respond promptly.
Search the InspectApedia website
Note: appearance of your Comment below may be delayed: if your comment contains an image, photograph, web link, or text that looks to the software as if it might be a web link, your posting will appear after it has been approved by a moderator. Apologies for the delay.
Only one image can be added per comment but you can post as many comments, and therefore images, as you like.
You will not receive a notification when a response to your question has been posted.
Please bookmark this page to make it easy for you to check back for our response.
Our Comment Box is provided by Countable Web Productions countable.ca
Citations & References
In addition to any citations in the article above, a full list is available on request.
- New York State Department of Health, APPENDIX 75-A WASTEWATER TREATMENT STANDARDS - INDIVIDUAL HOUSEHOLD SYSTEMS , [PDF] New York State Department of Health, 3 February 2010, retrieved 3/1/2010, original source: https://www.health.ny.gov/regulations/nycrr/title_10/part_75/appendix_75-a.htm
- "Bacterial Pathogens Recovered from Vegetables Irrigated by Wastewater in Morocco", Y. Karamoko, K. Ibenyassine, M. M. Ennaji, B. Anajjar, R. Ait Mhand, M. Chouibani, Journal of Environmental Health, June 2007.
Abstract:
The authors obtained 50 vegetable samples from various regions in Morocco and examined them to determine the micro biological quality of these products. Aerobic count, coliform, enterococci, and Staphylococcus areus were evaluated. This analysis revealed high levels of enterococci, fecal coliforms, and total coliforms. No coagulase-positive Staphylococcus aureas was detected in any of the samples analyzed. Biochemical identification of Enterobacteriaceae showed the presence of Citrobacter freundii (28 percent), Enterobacter cloacae (27 percent), Escherichia coli (16 percent), Enterobacter sakazakii (12 percent), Klebsiella pneamoniae (17 percent), Serratia liquefaciens (11 percent), and Salmonella arizonae (0.7 percent). The results clearly demonstrate that vegetables irrigated with untreated wastewater have a high level of microbiological contamination. Consequently, these vegetables may be a threat for the Moroccan consumer and may be considered a serious risk to Moroccan public health.ABSTRACT FROM AUTHOR Copyright of Journal of Environmental Health is the property of National Environmental Health Association and its content may not be copied or emailed to multiple sites or posted to a listserv without the copyright holder's express written permission. However, users may print, download, or email articles for individual use. This abstract may be abridged. No warranty is given about the accuracy of the copy. Users should refer to the original published version of the material for the full abstract.
Contact Us to request a copy of this article stored as BacterialPathogens.pdf if you have difficulty obtaining a copy of this full article for private use. At BACTERIAL PATHOGENS in FRUIT & VEGETABLES is a slightly-rough online copy from thefreelibrary.com - Beuchat, L.R. (1996). Pathogenic microorganisms associated with fresh produce. Journal of Food Protection, 59(2), 204-216.
- Breuer, T, Benkel, D.H., Shapiro, R.L., Hall. W.N., Winnett. M.M., Linn, M.J., Timothy, J.N., Barrett. J., Dietrich, S., Downes, EP, Toney, D-M., Pearson, J.L., Rolka. H., Slutsker. L, & Griffin, PM. (2001). A multi-state outbreak of Escherichia coli O157:H7 infections linked to alfalfa sprouts grown from contaminated seeds, Emerging Infectious Diseases, 7(6), 977-982.
- Evans, M.R., Ribiero, CD., & Salmon, R.L. (2003). Hazards of healthy Living: Boiled water and salad vegetables as risk Factors For Campylobacter infection. Emerging Infectious Disease, 9(10), 1219-1225.
- Guo, X., Chen, j . , Brackett, R.E., & Beuchat. LR. (2001). Survival of Saimonellae on and in tomato plants From the time of inoculation at flowering and early stages of fruit development through fruit ripening. Applied and Environmental Microbiology, 67(10), 4760-4764
- Madden, J.M. (1992). Microbial pathogens in fresh produce—The regulatory perspective. Journal of Food Protection, 55, 821-823.
- Shearer, A.E., Strapp, CM., & Joerger, R.D. (2001). Evaluation of polymerase chain reaction-based system for detection of Salmonella enteritidis, Escherichia coli O157:H7, Listeria spp., and Listeria monocytogenes on Fresh Fruit and vegetables. Journal of Food Protection, 64(6), 788-795.
- "Septic Tank/Drainfield System Fact Sheet", Utah Department of Environmental Quality, Division of Drinking Water, Source Protection Program - (801) 536-4200 Division of Water Quality - (801) 538-6146 Sonja Wallace, Pollution Prevention Coordinator - (801) 536-4477 Environmental Hotline - 1-800-458-0145 - Original source: http://www.drinkingwater.utah.gov/documents/spec_services/pollution_prevention_septic_tanks.pdf
- New York State Wastewater Treatment Standards - Individual Household Systems, Appendix 75-A (1990), Public Health Law 201(1)(1).
- New York State Alternative Septic System Design Regulations, Wastewater Treatment Standards - Individual Household Systems, Appendix 75-A, of Public Health Law 201(1)(1) 1990.
- Advanced Onsite Wastewater Systems Technologies, Anish R. Jantrania, Mark A. Gross, CRC Taylor & Francis, 2006 ISBN 0-8493-3029-7
- Anish Jantrania, Ph.D., P.E., M.B.A., Consulting Engineer, Mechanicsville VA, 804-550-0389
- The Septic System Owners's Manual, Lloyd Kahn, Blair Allen, Julie Jones, Shelter Publications, 2000. ISBN 0-936070-20-X
- Onsite wastewater treatment systems, Bennette D. Burks & Mary Margaret Minnis. Textbook and reference manual on all aspects of onsite treatment. This is one of the best books we've reviewed on the subject, with an excellent balance of clear simple explanation and solid engineering. Topics: Soil & Site Selection, Hydraulics, System Selection & Design, Wastewater Biology, History & Mythology of Onsite Wastewater Treatment. $49.95, Hogarth House, Ltd., 800-993-2665 x327 or order by telephone 800 -993-2665 x327 (Univ. Wisc. Bookstore)
- Mark Cramer Inspection Services Mark Cramer, Tampa Florida, Mr. Cramer is a past president of ASHI, the American Society of Home Inspectors and is a Florida home inspector and home inspection educator. Contact Mark Cramer at: 727-595-4211 mark@BestTampaInspector.com 11/06
- Thanks to reader Cheryl Sweetland for email discussion of planting fruit trees (avocado & mango trees) near a septic tank 07/13/2010
- Advanced Onsite Wastewater Systems Technologies, Anish R. Jantrania, Mark A. Gross. Anish Jantrania, Ph.D., P.E., M.B.A., is a Consulting Engineer, in Mechanicsville VA, 804-550-0389 (2006). Outstanding technical reference especially on alternative septic system design alternatives. Written for designers and engineers, this book is not at all easy going for homeowners but is a text I recommend for professionals--DF.
- In addition to citations & references found in this article, see the research citations given at the end of the related articles found at our suggested
CONTINUE READING or RECOMMENDED ARTICLES.
- Carson, Dunlop & Associates Ltd., 120 Carlton Street Suite 407, Toronto ON M5A 4K2. Tel: (416) 964-9415 1-800-268-7070 Email: info@carsondunlop.com. Alan Carson is a past president of ASHI, the American Society of Home Inspectors.
Thanks to Alan Carson and Bob Dunlop, for permission for InspectAPedia to use text excerpts from The HOME REFERENCE BOOK - the Encyclopedia of Homes and to use illustrations from The ILLUSTRATED HOME .
Carson Dunlop Associates provides extensive home inspection education and report writing material. In gratitude we provide links to tsome Carson Dunlop Associates products and services.